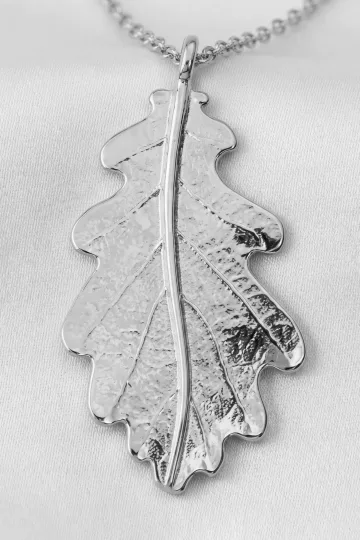
Sterling Silver 925: The Complete Guide to the World's Most Popular Alloy
Introduction: Why 925 Silver Is the Industry Standard
925 silver, also known as sterling silver, is the most sought-after material in modern jewellery making. It is not simply one of many options - it is a recognised global standard, used from small independent workshops to the largest fashion houses in the world.
Its popularity is no accident. Over centuries of use, this alloy has proven its versatility, reliability and aesthetic value. It combines the nobility of a precious metal with the practicality required for everyday wear.
Where 925 Silver Is Found
In the small workshops of independent designers
Young jewellers and independent artisans choose 925 silver as their primary material for experimentation. The reason is straightforward: it is accessible enough to allow the creation of complex, unique pieces without catastrophic financial risk. At the same time, it is prestigious enough to attract discerning buyers.
The metal forgives beginners' mistakes (it can be remelted and started over), yet demands mastery to create truly outstanding work. It is the ideal material for learning and growth.
In the collections of the world's greatest fashion houses
The leading jewellery and design houses create a significant portion of their collections in 925 silver. For them, this is not about cost-saving - it is a deliberate choice of a material that perfectly conveys their design philosophy.
The metal possesses a particular visual "temperature" that is unattainable with gold. It is more contemporary, less conservative, freer in interpretation.
In private collections of jewellery art
Museums and private collectors preserve masterpieces created centuries ago. Victorian brooches, Art Nouveau pendants, Art Deco bracelets - many iconic works of jewellery art are crafted in sterling.
This demonstrates the material's longevity. With proper care, 925 silver outlasts generations, preserving its beauty and value.
In mass jewellery production worldwide
925 silver is the foundation of the fashion jewellery industry. Millions of pieces are produced annually in India, China, Thailand, Italy and Turkey. This makes it the most accessible precious metal for the general public.
Its democratic nature does not diminish the value. It is a noble metal that virtually anyone can afford.
Why Buyers Choose Silver
Cool, pure lustre
Silver possesses a unique visual quality - its lustre is cooler and brighter than that of any other metal. It is not the warm glow of gold, nor the restrained mattness of platinum. It is a pure, almost icy light that instantly draws attention.
Properly polished, it reflects light better than any other metal (the highest reflectance coefficient among all metals - 95-99%). This makes it indispensable for pieces where the play of light is paramount.
Versatility
The metal suits any wardrobe, any style, any occasion. It does not flaunt status like gold, yet it does not disappear like costume jewellery. It is the balance between value and restraint.
Sterling jewellery is equally appropriate:
- In a professional office look
- In everyday casual style
- At an evening event
- In a creative, bohemian setting
- In a sporty urban look
This versatility makes it the ideal "foundation" of a jewellery wardrobe.
Affordability
The material costs tens of times less than gold and hundreds of times less than platinum. This allows one to:
- Buy more pieces, building a varied collection
- Experiment with styles without fear of "spending too much"
- Change jewellery to match mood and outfit
- Afford designer, unique pieces
- Give quality gifts without breaking the bank
Affordability does not mean cheapness. It means the democratisation of luxury.
Ability to change looks frequently
Since sterling jewellery is more accessible, one can have several sets for different occasions. Bold rings for everyday. Delicate earrings for the office. Statement necklaces for the evening.
Gold is often perceived as a "lifetime investment" - a single ring worn for decades. Sterling gives the freedom to change, try, play with style.
What the 925 Hallmark Means: Chemistry and Mathematics
The 925 hallmark is not merely a number stamped on metal. It is precise information about the alloy's chemical composition, its properties and characteristics.
The Metric Purity System
The hallmarking system is based on parts per thousand (millesimal fineness). The number 925 means that in 1,000 grams of alloy there are:
925 grams (92.5%) of pure silver
This is the foundation of the alloy. It is the precious metal that gives the material its characteristic colour, lustre and antibacterial properties. A noble element, valued by humanity for millennia.
The chemical symbol for silver is Ag (from the Latin Argentum). Atomic number 47. It is a transition metal with unique properties.
75 grams (7.5%) of alloying additives
This portion is called the "alloy" or "ligature." Most commonly it is copper (Cu), though theoretically other metals may be used (zinc, nickel in cheap alloys).
The alloy does not simply "dilute" the precious metal. It fundamentally alters the physical properties, making it suitable for use in jewellery.
Why Precisely 92.5% and Not 90% or 95%?
This percentage is the result of centuries of empirical experimentation by jewellers.
With a content below 92%:
- The alloy becomes too dark (copper begins to influence the colour)
- Reflective capacity worsens
- A noticeable yellowish or reddish tint appears
- Antibacterial properties weaken
With a content above 93%:
- The alloy becomes too soft for practical use
- Jewellery deforms rapidly
- It is impossible to create fine, openwork elements
- Stone setting becomes unreliable
92.5% is the sweet spot, refined over centuries of practice.
Historical Background: Where the 925 Standard Came From
The term "sterling silver" has deep historical roots.
Version 1: The German Merchants
According to one account, the name derives from "Easterling" - German merchants of the Hanseatic League who in the 12th century traded high-quality metal of a specific fineness with England.
Over time, "Easterling" was shortened to "Sterling."
Version 2: The Coins with a Small Star
Another version links the name to coins bearing a small star (star in English). "Starling" became "Sterling."
The Official Establishment of the Standard
In 1300, King Edward I of England legally established the standard for the metal in jewellery - 92.5% purity.
Since then, this standard has spread worldwide and become universal for the jewellery industry.
Why Additives Are Needed: The Role of the Alloy
Why can the pure metal (999 fineness) not be used in jewellery? The answer lies in understanding the physical properties of metals.
Pure Silver: Beautiful but Impractical
999 fineness (99.9% purity) is a virtually chemically pure element. It is used:
In investment bullion
Banks sell 999 bars as an investment asset. They are stored in vaults, not subjected to mechanical stress. For this purpose, the pure metal is ideal.
In commemorative coins
Collector coins are often made from 999 fineness. They are stored in capsules, not carried in pockets. The softness is irrelevant.
In industry
The pure form is used in electronics (high conductivity), in medicine (antibacterial properties), in mirror and photographic material production (high reflectance).
Why not in jewellery?
The pure metal is extremely soft and malleable. Its hardness on the Mohs scale is just 2.5-3 (for comparison: a human fingernail - 2.5, copper - 3, gold - 2.5-3).
This means:
It scratches easily
Even a rough fabric (denim, for example) can leave micro-scratches on the surface. After a few days of wear, a mirror polish transforms into a dull, scratch-covered surface.
It deforms under pressure
A pure ring can be bent with the fingers. Seriously. The pressure of the hand when clenching a fist is sufficient to deform the thin shank of a ring.
Pure chains stretch under their own weight. Bracelets lose their shape.
It cannot hold stones
The prongs (claws that hold the stone) in the pure form bend. The stone can fall out. An unacceptable risk for jewellery with precious settings.
It rapidly loses its polish
The pure form cannot be "polished" to achieve a long-lasting mirror finish. The surface quickly becomes dull and lifeless.
It turns into a shapeless mass
A jewellery piece made from the pure metal loses its original form within weeks of active wear. Crisp edges blur. Fine elements crush. The piece becomes a shapeless lump of metal.
Copper as the Ideal Additive
Of all possible alloying elements, copper has proven the best.
Why copper?
Compatible atomic lattice
The two elements are neighbours in the periodic table. Their atomic radii are close, allowing them to blend perfectly at the molecular level, forming a homogeneous alloy.
Does not affect the colour
7.5% copper is insufficient to alter the characteristic white colour. The 925 alloy is visually indistinguishable from the pure form.
Significantly increases hardness
Adding 7.5% copper increases the alloy's hardness approximately 2-2.5 times. Mohs hardness rises to 2.5-4.
This is sufficient to:
- Create fine, complex elements
- Make reliable prongs for stones
- Ensure long-term shape retention
- Maintain the mirror polish much longer
Preserves malleability
Despite increased hardness, the 925 alloy remains malleable and plastic. This is critical for jewellers who create pieces by hand, using forging, chasing and drawing techniques.
Availability and price
Copper is an inexpensive metal. It does not significantly increase the alloy's cost, unlike more exotic alloying additives.
Alternative Alloying Elements
Although copper is the standard, other additives are sometimes used.
Nickel
In cheap alloys, nickel is sometimes added. It increases hardness but has a serious drawback - high allergenicity.
Nickel is one of the most common causes of contact dermatitis. Many people develop an allergic reaction upon contact with nickel.
Quality manufacturers NEVER use nickel in the 925 alloy.
Zinc
Sometimes added in small quantities (alongside copper) to improve casting properties. Zinc lowers the melting point, making the alloy more fluid, improving mould fill during casting.
Platinum, Palladium
In exclusive premium-segment alloys, small quantities of platinum group metals are sometimes added. This improves anti-oxidation properties, slowing tarnish.
Such alloys are considerably more expensive and rarely used.
Why 925 and Not 999: A Comparative Analysis
Is the pure form (999 fineness) not better by definition? Let us examine the question in detail.
999 Silver: Characteristics and Applications
What it is: 99.9% pure silver, minimal impurities (less than 0.1%).
Where it is used:
Bank investment bullion
The 999 form is a traded commodity, a financial asset. Bars from 1 ounce to 1,000 ounces are purchased by investors as inflation protection and portfolio diversification.
They remain in vaults, do not suffer wear. The softness of the metal is irrelevant.
Commemorative and collector coins
Many modern commemorative coins are struck in 999 fineness. They are stored in capsules, not used as currency.
Historical coins were often of lower fineness (e.g. 500-900), because wear resistance was needed for circulation.
Decorative plating
In certain technologies (electroplating, vacuum deposition), the pure form is used to coat other metals.
Industrial applications
Electronics (solders, contacts), medicine (antibacterial coatings), mirror and photographic material production.
Why NOT in jewellery:
- Unsuitable for everyday rings - deforms under pressure
- Cannot support large stones - prongs bend
- Rapidly loses shape and polish - the piece becomes a worn lump of metal
- Impossible to create fine, delicate elements - they would simply bend
- Cannot hold engraving or carving - details "wash out"
925 Silver: The Optimal Balance
What it is: 92.5% silver + 7.5% copper (or other additives)
Where it is used:
Jewellery of every category
From simple earrings to complex statement necklaces. From fine chains to chunky men's signet rings. From classic wedding bands to avant-garde designer jewellery.
Table silver
Historically, 925 silver was used to make cutlery and tableware. Forks, spoons, knives, trays, teapots - all were made from sterling.
Durability was needed (tableware is used daily) as was beauty (a full service was a mark of status).
Decorative objects
Photo frames, candlesticks, boxes, figurines. Everything requiring the union of aesthetics and functionality.
Musical instruments
High-quality concert flutes are often made from the 925 alloy. The metal influences the sound, making it warmer and richer.
Advantages:
- Global standard for quality jewellery - recognised everywhere, from London to Tokyo
- High mechanical hardness - the piece holds its shape for decades
- Allows complex handwork - fine carving, filigree, chasing
- Lasts decades with proper care - these are pieces that cross generations
- The benchmark for work with enamels and stones - optimal base for any technique
The 925 alloy is a compromise, but a brilliant one:
Visually, it is indistinguishable from the pure form - the same vivid, cool, white lustre. Yet it is functional, practical and durable.
It is the perfect balance between beauty and strength.
Key Characteristics of 925 Silver: In Detail
Physical Properties
Density: 10.36 g/cm³ (for comparison: gold 19.3 g/cm³, platinum 21.45 g/cm³)
This means the material is considerably lighter than gold. A chunky ring weighs less than a gold ring of the same size. This makes large jewellery more comfortable to wear.
Melting point: approximately 893 °C (pure silver melts at 961 °C; copper lowers this temperature)
This is important for jewellers. A lower melting point makes working easier, reduces energy costs, decreases the risk of deformation during repairs.
Thermal conductivity: The highest of all metals (429 W/(m·K))
A ring quickly takes on body temperature. This makes it comfortable - no sensation of cold metal on the skin.
This same property is used to check authenticity: hold the piece in your hand - it will warm up within seconds.
Electrical conductivity: Also the highest among metals
Of no practical relevance to jewellery, but widely exploited in industry.
Hardness: 2.5-4 on the Mohs scale (depending on treatment and work-hardening)
Softer than steel (4-4.5), but harder than pure gold (2.5-3). Sufficient for jewellery, but requires careful handling.
Chemical Composition and Safety
Base: Silver (Ag) - 92.5%
A noble metal, it does not react with oxygen under normal conditions, does not corrode. It possesses antibacterial properties (the ions destroy bacterial cell walls).
Alloying additive: Most commonly copper (Cu) - 7.5%
Copper is also non-toxic and does not cause allergies in the vast majority of people. It is a safe metal, used by humanity for millennia.
Important: Nickel
In cheap, low-quality alloys, nickel may be used instead of (or alongside) copper. Nickel is considerably cheaper than copper, which tempts unscrupulous manufacturers.
The problem: Nickel is one of the most allergenic metals. Statistically, nickel allergy affects 10-20% of the population (especially women).
Symptoms of nickel allergy:
- Redness of the skin at the point of contact
- Itching, burning
- Appearance of a fine rash
- In severe cases - weeping eczema
How to avoid it:
- Buy jewellery from reputable brands
- Request the alloy composition certificate
- Avoid suspiciously cheap pieces
- If allergic - choose rhodium-plated silver or hypoallergenic materials
Quality 925 silver is hypoallergenic
With the correct composition (silver + copper, no nickel), the alloy is absolutely safe even for the most sensitive skin.
Silver even has a mild antiseptic effect - silver ions inhibit bacterial growth. This makes it ideal for earrings (contact with the piercing).
Mechanical Strength: What Can Be Created
925 silver opens the widest possibilities for jewellers to realise the most complex designs.
Fine openwork weaves
Complex chain links (Bismarck, Venetian, Python, Foxtail) require the metal to be strong enough to hold its shape, yet plastic enough for complex deformation.
925 silver is perfect for this. Links less than a millimetre thick hold their shape, do not open, do not deform during wear.
Chunky men's rings
Large signet rings with sharp edges, precise lines require a hard metal. A soft metal would "blur" the edges; sharp corners would round off.
925 silver maintains crisp geometry, preserves detail.
Complex clasps
Clasps for bracelets and necklaces, butterfly backs for earrings, lobster claws - all these mechanisms require precision and strength.
925 silver is hard enough for spring mechanisms to function for decades without losing their elasticity.
Reliable stone prongs
Prongs (the claws that hold the stone in the setting) must be strong enough not to bend. At the same time, they must be thin so as not to obscure the stone's beauty.
925 silver allows the creation of prongs 0.5-0.7 mm thick that hold the stone securely.
Detailed carving and engraving
Fine lines, small details, complex ornaments require the metal not to "flow" under the graver. 925 silver maintains the sharpness of carving.
Filigree
An ancient technique in which the finest silver wires (0.2-0.4 mm) are twisted, curved, soldered into openwork patterns. It requires a perfect balance of plasticity and strength.
925 silver is the classic material for filigree.
Chasing and embossing
Creation of relief patterns by pressing the metal. Requires plasticity (the metal must deform) but also strength (to hold the form afterwards).
925 silver is ideal for these techniques.
Aesthetic Properties
Colour
The 925 alloy has a characteristic white colour with a cool, slightly bluish tone. It is not the yellow of gold, nor the grey of steel - it is a unique, unmistakable lustre.
This colour is universal. It suits any skin tone, pairs with any clothing colour.
Lustre
The metal has the highest reflectance coefficient among all metals - up to 95-99%. For comparison: gold - 85-95%, platinum - 60-70%.
This means a properly polished piece shines brighter than any other metal. Its lustre is cool, pure, almost mirror-like.
Ability to patinate (blacken)
A unique property of this precious metal is its capacity for controlled oxidation (blackening). This is used to create an artistic effect.
The craftsperson applies a special solution (most commonly sodium sulphide) to the piece. The surface darkens. Then the raised portions are polished - they become bright, while the recesses remain dark.
This creates contrast, highlights relief, adds volume and a "vintage" feel to the piece.
The blackening technique is used in:
- Slavic jewellery traditions (ancient Russian techniques)
- Eastern jewellery (Indian, Thai silver)
- Gothic and biker jewellery
- Vintage and antique styles
Absence of "yellowing"
Unlike gold, the metal has no warm yellow tone. This makes it ideal for:
- Contemporary minimalist designs
- Cool, "cosmic" aesthetics
- Pairing with white and cool stones (diamonds, topazes, amethysts)
- Industrial, urban style
Practicality and Daily Wear: A Detailed Breakdown
Can 925 silver be worn every day, without ever removing it? Yes, but with an understanding of the material's characteristics and some sensible rules.
925 Silver Rings
Rings are the most popular type of jewellery, but also the most susceptible to wear. They are constantly in contact with surfaces, subjected to pressure and friction.
Shank thickness - a critical parameter
The shank is the base of the ring, the part that encircles the finger. Its thickness determines durability.
Thin shank (less than 1.5 mm):
Pros:
- Delicate, lightweight appearance
- Minimal weight
- Comfort (not felt on the finger)
Cons:
- Easily deformed by hand pressure
- May take on an oval shape (instead of round)
- Higher risk of fracture at the solder zone
- Requires periodic reshaping by a jeweller
- Unsuitable for large stones (the base cannot support the weight)
Optimal thickness: for women's rings - 1.5-2.5 mm, for men's rings - 2-4 mm
Medium shank (1.5-2.5 mm for women's, 2-4 mm for men's):
Optimal balance. The ring holds its shape, looks respectable, lasts a long time.
Thick shank (over 2.5 mm for women's, over 4 mm for men's):
Maximum strength. Virtually indeformable. Suitable for chunky statement rings.
May feel heavier, takes up more space on the finger.
Construction type
Cast rings (created by casting in a mould) are stronger than stamped ones. The metal is homogeneous; there are no soldered joints, which are zones of weakness.
Forged rings (created by deforming a blank) exhibit work-hardening - strengthening of the metal through mechanical processing. These are the strongest rings.
Wearing frequency and conditions
Daily wear at the office, at home: Sterling rings handle this perfectly at the right thickness.
Intense physical work, the gym: Best to remove them. Risk of deformation, scratches.
Contact with chemicals: Remove without fail. Chlorine and acids damage the surface.
925 Silver Earrings
Earrings are the ideal piece for the 925 alloy.
Why the material is perfect for earrings:
Minimal load
Earrings hang from the earlobe or are threaded through the piercing. They are not subjected to friction against surfaces (like rings), nor to pressure (like bracelets).
This means that even delicate, fine earrings last decades without deforming.
Antiseptic properties of silver
The ions have an antibacterial effect. Especially important for earrings, which are in contact with the piercing - a potential infection zone.
The 925 alloy reduces the risk of piercing inflammation, particularly for those who have recently had their ears pierced.
Hypoallergenicity
Quality 925 (without nickel) does not cause allergies. Critical for earrings, which are worn for months without removal.
Variety of designs
From minimalist studs weighing fractions of a gram to chunky statement earrings with drops. The 925 alloy allows any idea to be realised.
Earring types and their characteristics:
Studs: Small, flush against the lobe. Can be worn constantly, even to sleep. Virtually not subject to wear.
Butterfly back (push back): Reliable construction. The mechanism may loosen over time, but is easily tightened by a jeweller.
Hook (shepherd's hook): Elegant but less secure. Higher risk of losing the earring. Suitable for not-too-heavy earrings.
Hoops: May deform under strong pressure (e.g. if stepped on). Remove at night.
Long drop earrings: May tangle in hair. Drops may bend. Remove before physical activity.
Chains and Bracelets
Chains and bracelets are jewellery with a high risk of breakage. They consist of many links connected to one another. Each link is a potential point of failure.
Link types and their strength:
Cable link: Simple; each link perpendicular to the previous one. Fairly strong, but may twist.
Curb link: Links in the same plane, closely fitted. Stronger than cable; tangles less.
Bismarck: Complex weave of many small elements. Very strong, chunky. Suitable for men's chains and bracelets.
Venetian (box chain): Square or rectangular links. Elegant, strong. Universal.
Snake: Smooth, resembling snakeskin. Flexible, but can kink at one point, leading to breakage.
Singapore: Twisted weave, sparkling in the light. Medium strength. May twist.
Rules for wearing:
Remove before sport
Any physical activity involving sudden movements (running, jumping, pull-ups) stresses the chain. Links stretch; at the weakest point, a break occurs.
Check the clasp
The clasp is the weakest point of a chain or bracelet. Spring mechanisms wear out. Check that the clasp closes securely before each outing.
If the clasp is loose or does not fix properly - take it to a jeweller for repair. Repairing a clasp costs little. Losing a beloved piece of jewellery - priceless.
Do not pull
If the chain catches on clothing - do not pull. Carefully untangle it. A sudden jerk can snap a link.
Do not hang pendants that are too heavy
If the pendant weighs more than the chain itself - it is a risk. The weight stretches the links, especially the central ones (which bear the maximum load).
Repairing silver chains:
A broken chain is not a death sentence. A jeweller can:
- Solder the broken link
- Replace the damaged section
- Shorten the chain if the damage is irreparable
Why Silver Tarnishes: Chemistry and Myths
The tarnishing of silver (patination) is surrounded by many myths. Let us see what actually happens. If your piece has already darkened and you want a step-by-step rescue rather than the chemistry, jump to the dedicated guide on what to do when jewelry has tarnished.
The Chemistry of the Oxidation Process
Myth: Silver tarnishes due to oxidation by atmospheric oxygen.
Reality: The metal barely reacts with pure oxygen under normal conditions. It tarnishes due to a reaction with sulphur compounds.
Chemical reaction:
2Ag + H₂S → Ag₂S + H₂
Silver + hydrogen sulphide → silver sulphide + hydrogen
The resulting compound (Ag₂S) is a dark, almost black substance that forms a film on the metal's surface.
Where the sulphur comes from:
In the air: Especially in industrial cities. Factory emissions, car exhausts contain sulphur compounds (SO₂, H₂S).
In clean mountain or sea air, the metal tarnishes much more slowly.
In human perspiration: Sweat contains various chemical compounds, including sulphur-containing amino acids (cysteine, methionine).
Different people have different sweat chemistry. For some, the metal tarnishes in a week; for others, in a year. It is individual.
In cosmetics and perfume: Many creams, lotions and perfumes contain sulphur-based preservatives or ingredients. Contact with them accelerates tarnishing.
In household chemicals: Cleaners, bleach, washing-up liquid - many contain sulphur or its compounds.
In certain foods: Eggs (especially the yolk), onion, garlic, legumes contain sulphur. If you cook while wearing a silver ring - it will blacken quickly.
It is important to understand:
The sulphide (tarnish) is a surface film just a few microns thick. It does NOT penetrate deep into the metal, does NOT destroy its structure, does NOT reduce its weight or volume.
It is a purely aesthetic defect, easily removed by cleaning.
Tarnishing - Proof of Authenticity
Myth: "Real silver does not tarnish."
Reality: It is exactly the opposite. If the metal does NOT tarnish - there is reason to doubt its authenticity.
Rhodium-plated pieces (coated with a layer of rhodium) do not tarnish, because rhodium does not react with sulphur. But beneath the rhodium lies the real metal, which will tarnish if the coating wears off.
If a piece is sold as silver, is not rhodium-plated, but does not tarnish for years - it is probably not genuine. It may be nickel silver (a copper-nickel alloy), German silver (a copper-nickel-zinc alloy), or outright costume jewellery alloy.
Tarnishing Speed: What Affects It
Factors that accelerate tarnishing:
- Air humidity: The higher the humidity, the faster the reaction. In dry climates, tarnishing is slower.
- Air pollution: Industrial cities, proximity to roads - more sulphur in the air.
- Skin contact: Individual sweat chemistry. In people with more acidic sweat, the metal tarnishes faster.
- Contact with cosmetics: Creams, perfumes, hairsprays accelerate tarnishing.
- Contact with rubber: Yes, rubber (gloves, hair bands) contains sulphur and causes rapid blackening.
- Storage: Exposed to open air, it tarnishes faster than in a closed jewellery box.
Factors that slow tarnishing:
- Dry, clean air
- Storage in airtight bags or jewellery boxes
- Minimal contact with skin and cosmetics
- Rhodium plating
- Regular cleaning (removing the initial film before it becomes thick and dark)
How to Slow Tarnishing: Practical Tips
Rhodium plating
Rhodium is a platinum group metal (like platinum, palladium, iridium). It does not tarnish, does not oxidise, does not react with sulphur.
The rhodium plating process:
The jewellery piece is immersed in a galvanic bath containing a rhodium solution. Under the action of electric current, a thin layer of rhodium is deposited on the silver surface (0.1-0.25 microns).
Advantages of rhodium plating:
- Does not tarnish: The piece maintains a bright white lustre for years.
- Cool "steel" lustre: Rhodium gives the metal a distinctive, cool tone that many find more modern and stylish than the "warm" natural finish.
- Scratch protection: Rhodium is harder than the base metal; the coating protects against minor scratches.
- Hypoallergenicity: Rhodium is absolutely hypoallergenic - important for people with sensitive skin.
Disadvantages of rhodium plating:
- Temporary: The rhodium coating wears off. Lifespan - from 6 months to 3-5 years depending on wear intensity and coating thickness.
- Requires renewal: When the rhodium wears off (usually unevenly, creating "bare patches"), re-plating is needed.
- Changes the tone: Rhodium-plated pieces look different from the natural metal. If you prefer the "warm" lustre - rhodium plating is not for you.
- Not applicable to blackened pieces: Rhodium plating destroys the blackening (patina).
Proper storage:
- In a closed jewellery box or pouch (limit air contact)
- Separately from other jewellery (avoid scratches)
- In a dry place (not in the bathroom)
- A piece of chalk or silica gel sachet in the bag helps (they absorb moisture)
Minimise contact:
- Put jewellery on AFTER applying cosmetics and perfume
- Remove before cleaning, cooking
- Remove before sport (perspiration)
- Remove before sleeping
Regular wiping:
Even if the piece has not yet visibly tarnished, an invisible film forms on it. Regular wiping (once a week) with a special polishing cloth or soft fabric prevents the build-up of a thick sulphide layer.
How to Choose Quality 925 Silver Jewellery
The sterling jewellery market is vast - from street stalls to exclusive boutiques. How can you avoid mistakes and choose a genuinely quality piece?
Checking the Hallmark (Fineness)
The hallmark is the official confirmation of the metal's fineness.
Where to look for the hallmark:
On rings: Inside the shank. Sometimes the hallmark is so small a loupe is needed.
On earrings: On the clasp, on the post (for studs), on the inner side of the element.
On chains and bracelets: On the clasp or on the jump ring next to the clasp.
On pendants and charms: On the bail (the loop through which the chain passes) or on the reverse.
What should be on the hallmark:
In the United Kingdom:
- The number 925 - the fineness
- The assay office mark (anchor for Birmingham, leopard's head for London, rose for Sheffield, castle for Edinburgh)
- The sponsor's or maker's mark
In other countries:
- USA: "925", "Sterling", "Sterling Silver"
- Europe: "925" or country-specific symbols
Absence of hallmark:
If a piece has NO hallmark - this does NOT necessarily mean it is fake. It may be:
- A handmade piece by an independent craftsperson
- An antique (old pieces may have had different marking systems, or the hallmark has worn off)
- A foreign piece with a hallmark different from the local one
- Genuinely a fake or costume jewellery
How to verify: If the seller claims it is 925 silver but there is no hallmark - ask for a certificate or assay. A responsible seller will provide one.
Weight and Thickness - Crucial Parameters
The metal is fairly dense. If a piece looks chunky but weighs suspiciously little - it is probably hollow inside (which is not necessarily bad) or is not genuine sterling.
Weight check:
Ask to have the jewellery weighed. Sellers in jewellery shops always have precision scales.
Approximate reference weights:
- Thin women's ring: 2-4 grams
- Medium women's ring: 4-8 grams
- Chunky men's ring: 10-25 grams
- Fine chain (40-45 cm): 5-10 grams
- Medium chain: 10-20 grams
- Heavy chain: 25-60+ grams
- Small studs: 1-3 grams per pair
- Medium earrings: 5-10 grams per pair
- Large earrings: 15-30+ grams per pair
If the weight is significantly lower than expected:
- The piece may be hollow - not always negative, but you should know
- The metal thickness may be too thin - risk of rapid deformation
- It may not be 925, but a lighter metal (aluminium, low-content alloys)
Thickness check:
For rings: Look at the shank from the side. If it is thinner than 1.5 mm - it is for occasional wear. For daily use, you need at least 1.5-2 mm.
For chains: Overly thin links (wire diameter under 0.5 mm) break easily. The optimum is 0.7-1.5 mm depending on the link type.
For decorative elements: If an element looks voluminous but is clearly hollow inside (you can feel it with your fingers or by gentle pressure) - it is a hollow construction. Lighter and less expensive, but less durable.
Quality of Finish
The quality of polishing and finishing is a clear indicator of manufacturing level.
Surface check:
Inspection under bright light:
Hold the piece near a bright light source (lamp, window). Turn it at various angles.
What to look for:
- Pores, "bubbles": If small cavities or pores are visible - it is a casting defect. The metal was insufficiently degassed; air remained in the mould. Such a piece is less durable.
- Scratches: On a new piece, there should be no scratches. Their presence indicates poor packaging, transport, or that the piece has been previously worn.
- Uneven lustre: Quality polishing produces a mirror-like, uniform surface. If there are matt patches, streaks - the polishing is poor.
- Tool marks: There should be no visible marks from polishing discs, sandpaper, files.
Checking joints and seams:
If the piece is composite (e.g. stone in setting, elements soldered together):
- Solder joints: Should not be visible. Quality soldering is invisible. If the joint is apparent, raised, or a different shade - it is a sign of cheap manufacture.
- Prongs (for stones): All prongs should be the same length, symmetrically bent, pressing the stone firmly. If prongs are uneven, crooked, or the stone wobbles - it is poor work.
- Setting: The stone should not move. Check with your finger - try to nudge the stone slightly. If it moves - the setting is unreliable, the stone could fall out.
Checking clasps:
For chains and bracelets:
- Open and close the clasp several times. It should work smoothly and lock securely.
- For lobster claws - the spring should be springy, the tongue should close firmly.
- For spring rings - the ring should be tight enough not to open too easily.
For earrings:
- Butterfly backs should click into place with a light snap, hold firmly.
- Hooks should have a sufficiently rigid curve so the post does not slip out.
- Clasps should be symmetrical on both earrings.
Reputation of the Manufacturer/Seller
Buying from a well-known brand:
Pros:
- Quality guarantee (they value their reputation)
- Certificates, documentation for the piece
- After-sales service (cleaning, repair)
- Return/exchange possibility
Cons:
- Brand markup
- Less unique designs (mass production)
Buying from an independent maker:
Pros:
- Unique, exclusive designs
- Often - better value for money
- Possibility of bespoke orders
- Personal, attentive approach
Cons:
- Need to carefully check the portfolio and reviews
- Fewer guarantees (though good makers do offer guarantees)
- May not have an official hallmark
Buying from high street retailers:
Pros:
- Accessible prices
- Wide selection
- Convenience (can buy at the shopping centre)
Cons:
- Often - lower quality (thin metal, poor polishing)
- Standard designs
- Minimal after-sales service
How to check reputation:
- Read reviews (Google, Trustpilot, social media)
- Check how long the company/maker has been operating
- Verify the existence of a physical address (not just a website)
- Ask friends who are interested in jewellery
- Check the portfolio (for makers) - photo quality, variety of work
Common Buyer Mistakes: What to Avoid
Mistake No. 1: Expecting "Eternal Whiteness"
Myth: "I'll buy it, and it will always be white and shiny."
Reality: Sterling is a "living" metal. It interacts with its environment, changes, patinates.
What to do:
Option A: Accept and appreciate
Many enthusiasts consider a light patina part of the charm. The vintage, slightly darkened look gives the piece character, history.
Option B: Rhodium plate
If you categorically do not wish to bother with cleaning - choose rhodium-plated pieces. They will retain their whiteness for years.
Option C: Regular cleaning
Accept cleaning as a care ritual. Once or twice a month - 10 minutes, and the piece shines anew. It is part of the culture of owning fine jewellery.
Mistake No. 2: Sleeping in Jewellery
Problem:
Many wear their jewellery round the clock, not removing it even at night. This leads to:
Deformation:
During sleep, you toss and turn. The ring can end up under your body weight, bending. The chain can tangle, pull taut, and a link snaps.
Accelerated wear:
Friction against bedding and pillows accelerates the wearing down of the polish.
Discomfort:
Jewellery can disturb sleep, dig into skin, catch in hair.
Risk of injury:
Earrings can dig into the skin when sleeping on one's side. A chain can tighten around the neck (a theoretical risk, but it exists).
What to do:
ALWAYS remove your jewellery before sleeping.
Build the habit: arrive home → remove jewellery → place in box.
It will take 30 seconds but extend the life of your jewellery enormously.
Mistake No. 3: Contact with Aggressive Chemicals
Problem:
Chlorine, acids, alkalis in cleaning products damage silver.
Chlorine:
Bleach, toilet cleaners, swimming pool chlorine - all of these are a CATASTROPHE for silver.
Chlorine reacts with silver, forming silver chloride. The surface becomes matt, porous, "eaten away." This is irreversible damage.
Sulphur-containing products:
As we already know, sulphur causes rapid blackening.
Ammonia, peroxide:
Aggressive to silver; may cause discolouration.
What to do:
Remove rings before:
- Washing dishes
- Household cleaning
- Any work with chemicals
Wear gloves:
If you must work with chemicals and do not wish to remove your jewellery - rubber gloves. But remember: rubber itself contains sulphur, so it is not an ideal long-term solution.
Do not swim in pools wearing silver:
Chlorinated water is silver's enemy. Remove jewellery before swimming.
Mistake No. 4: Ignoring Individual Reactions
Problem:
In some people, the metal tarnishes VERY quickly. In a single day of wear, a visible patina may appear.
Cause:
Individual sweat chemistry. In people with more acidic perspiration, with a certain microelement balance, it tarnishes faster.
This does not mean something is wrong with the person. It is normal variability.
What to do:
- Clean your jewellery after each wearing
- Consider rhodium-plated pieces
- Rotate your jewellery (do not wear the same piece every day)
- Accept it as a characteristic; do not worry
Mistake No. 5: Storing in the Bathroom
Problem:
The bathroom is the most humid room in the house. Humidity accelerates all chemical reactions, including tarnishing.
What to do:
Store your pieces in the bedroom, in a jewellery box. Not in the bathroom.
Detailed Guide to Home Care for 925 Silver
Cleaning the metal requires neither major investment nor complex procedures. It can be done at home, effectively and safely. If you also own gold pieces, the complete guide to cleaning gold and silver at home covers both metals side by side, plus what to do (and not do) with gemstones.
Method 1: Soapy Solution (Regular Cleaning)
Purpose: Removal of grease, dust, light patina.
What you need:
- Warm water (not boiling!)
- Mild soap or washing-up liquid (without abrasives)
- Soft toothbrush or cotton buds
- Soft cloth (microfibre)
Process:
- Pour warm water into a bowl
- Add a few drops of soap, stir
- Place the piece in the solution for 5-10 minutes
- With the soft brush, gently clean (especially hard-to-reach spots - between links, under stones)
- Rinse with clean water
- Dry with a soft cloth
Pros: Safe, gentle, suitable for frequent use.
Cons: Will not tackle heavy tarnishing.
Method 2: Special Silver Cloths
What they are: Special cloths impregnated with a solution that dissolves the sulphide tarnish.
Where to buy: Jewellery shops, online.
How to use:
- Remove the cloth from its packaging
- Rub the jewellery (the cloth will turn black - this is normal; dissolved sulphide remains on it)
- If needed - wipe with a clean, dry cloth for a final polish
Pros:
- Very fast (30 seconds per piece)
- Effective against tarnishing
- Convenient (no water needed; can be done anywhere)
- Cleans and polishes simultaneously
Cons:
- Cost (cloths need to be purchased regularly)
- Not suitable for pieces with porous stones (turquoise, pearl, coral - the cloth's solution may damage them)
Method 3: Special Silver Solutions
What they are: Liquids for soaking sterling pieces. They dissolve sulphide tarnish, restoring lustre.
Where to buy: Jewellery shops, hardware stores.
How to use:
- Pour the solution into a bowl (glass or plastic, NOT metal)
- Place the piece in the solution
- Soaking time - from 30 seconds to 5 minutes (read the instructions on the specific product)
- Remove, rinse with water
- Dry with a soft cloth
Pros:
- Very effective against heavy tarnishing
- Works quickly
- Penetrates all hard-to-reach areas
Cons:
- Aggressive (may damage some stones, pearls, enamels)
- Unpleasant smell
- The time must be strictly observed (exceeding it may leave the metal matt)
Method 4: Silver Paste
What it is: A cream-like paste for polishing sterling jewellery.
How to use:
- Apply the paste to a soft cloth
- Rub over the piece in circular motions
- Pay attention to tarnished areas
- Rinse with water
- Dry
Pros:
- Effective
- Cleans and polishes simultaneously
Cons:
- Labour-intensive (requires active rubbing)
- Not suitable for pieces with complex relief (paste accumulates in recesses)
Home Methods (With Caution!)
Bicarbonate of soda + Aluminium foil + Boiling water:
Principle: The chemical reaction reduces silver from the sulphide.
Process:
- Line the bottom of a bowl with aluminium foil
- Sprinkle bicarbonate of soda (2-3 tablespoons)
- Place the jewellery on the foil
- Pour boiling water over it
- Wait 5-10 minutes (a reaction with a hydrogen sulphide smell should occur)
- Remove, rinse, dry
Pros: Effective, cheap.
Cons:
- Dangerous for stones: Boiling water can damage many stones (opals may crack, pearls cloud, enamels degrade)
- Unpleasant smell: Hydrogen sulphide is released
- Risk of damaging the metal: If left too long, it may become matt, porous
WARNING: Use this method ONLY for plain jewellery without stones or enamels!
What You Should ABSOLUTELY NOT Do
Toothpaste:
Why not: It contains abrasive particles (silicon dioxide, calcium carbonate). These microscopic particles scratch the surface.
With each toothpaste cleaning, more and more micro-scratches appear on the surface. Over time, the mirror polish becomes a matt, scratched surface.
Bicarbonate of soda as an abrasive:
Why not: Bicarbonate of soda consists of crystals. Applied dry and rubbed, it acts like sandpaper, scratching the metal.
You can: Use bicarbonate of soda in DISSOLVED form (in water) for soaking. Never rub with dry bicarbonate.
Aggressive acids and alkalis:
Why not: They can destroy the metal surface, making it matt, porous. Especially dangerous for stones.
Hard brushes, scouring pads:
Why not: They scratch the surface.
Boiling pieces with stones:
Why not: Many stones cannot withstand thermal shock:
- Opals: May crack, lose their play of colour
- Emeralds: Often filled with oil to enhance appearance; boiling water washes the oil out
- Pearls: Organic, deteriorate at high temperatures
- Amber: Also organic, may cloud
- Turquoise: Porous, may change colour
Ultrasonic cleaning for pieces with fragile stones:
Ultrasonic cleaning creates microvibrations. If the stone has internal fractures (and many natural stones do), ultrasound can widen them, causing the stone to crack.
Jewellery Storage: Rules and Tips
Proper storage is half the battle in maintaining the beauty of your pieces.
The Fundamental Rules of Storage
Dry location:
Humidity is the enemy. The drier the air, the slower the tarnishing.
Dark location:
Direct sunlight does not harm the metal directly, but accelerates some chemical reactions.
Separate storage:
Each piece - separately. This prevents:
- Scratches (hard stones scratch metal)
- Tangling (chains tangle with each other)
- Deformation (a heavy piece can crush a light one)
Airtightness (desirable):
Less contact with air means slower tarnishing. Ideal: individual zip-lock bags.
Storage Options
Compartmented jewellery box:
The classic option. Choose a box with:
- Soft lining (velvet)
- Multiple compartments
- Specific holders for rings, earrings
- A tightly closing lid
Pros: Beautiful, practical, everything in one place.
Cons: Jewellery is still in contact with air.
Individual pouches:
Soft fabric pouches (usually included with the jewellery purchase).
Pros:
- Each piece is isolated
- Fabric absorbs minimal moisture residue
- Compact
Cons: Less aesthetic than a box.
Zip-lock bags:
Transparent bags with a seal.
Pros:
- Nearly airtight (minimal contact with air)
- Transparent (you can see what is inside)
- Inexpensive
Cons: Not very aesthetic.
Tip: Place a piece of chalk or a silica gel sachet (the sort found in shoe boxes) in the bag. They will absorb moisture, and the silver will tarnish even more slowly.
Hanging organiser:
Textile organiser with pockets, hung in the wardrobe.
Pros:
- Compact (does not take up shelf space)
- Convenient (everything visible)
- Good ventilation
Cons: Contact with air (tarnishes faster than in a closed box).
Where NOT to Store
In the bathroom:
Even if you have a lovely box on the bathroom shelf - bad idea. Post-shower humidity is extreme. The metal will blacken within months.
In sunlight:
Do not leave jewellery on the windowsill, on a chest of drawers by the window.
With rubber items:
Rubber contains sulphur. If you store pieces near rubber gloves or hair bands - they will blacken quickly.
Loose in a single box:
Everything will tangle, scratch and deform.
Storage While Travelling
Problem: When travelling, you don't have your usual jewellery box. How do you store and transport jewellery?
Solution:
Small travel jewellery case:
Special compact travel cases. Hard shell (protection against pressure in luggage), soft lining, compartments.
Roll-up organiser:
A soft organiser that rolls up. Saves space, protects against pressure.
Individual pouches:
Each piece in its own zip-lock or fabric pouch, all in one toiletries bag.
Rule: Do not take all your valuable jewellery on a single trip. Leave some at home. Risk of loss or theft.
925 Silver and Stones: Ideal Pairings
Sterling is a universal setting for precious and semi-precious stones. Its cool, neutral tone does not compete with the stone's colour but enhances it.
Why Silver Is the Best Choice for Many Stones
Neutrality:
The white colour adds no tint to the stone. Gold (especially yellow) can cast a warm reflection, altering the perception of the stone's colour. Sterling is neutral.
Modernity:
The metal is associated with modern, minimalist design. A stone in sterling looks more current, less "old-fashioned."
Affordability:
A sterling setting costs less than gold. This allows you to buy a larger or higher-quality stone, investing the main budget in the stone rather than the metal.
The Best Pairings
Rock crystal (Clear quartz):
The absolute clarity of crystal pairs perfectly with the cool lustre. Together they create an effect of ice, purity.
Rock crystal is inexpensive, making such jewellery very accessible.
Topaz (Blue, London Blue, Sky Blue):
The cool tones of blue topaz and sterling are a classic. The metal highlights the purity and depth of the blue.
Topaz is fairly hard (8 on Mohs), unafraid of daily wear.
Amethyst:
Purple amethyst in silver - a pairing beloved since Victorian times. The cool metal tempers the warmth of purple, creating balance.
Amethyst is accessible and varied (from pale lilac to deep violet).
Garnet:
Dark red garnet and white sterling - a contrast that has worked for centuries. The metal makes the red even more intense, dramatic.
Onyx (Black):
Black onyx and white sterling - monochrome elegance. This pairing is popular in men's jewellery.
Onyx symbolises protection and strength.
Turquoise:
A classic pairing, especially in ethnic and bohemian styles. The vivid turquoise colour contrasts with the cool white metal.
Important: turquoise is porous and requires gentle care.
Moonstone:
The milky white moonstone with its mystical bluish shimmer (adularescence) suits sterling perfectly. Both materials are associated with the moon, magic, feminine energy.
Pearls:
White or cream pearls and sterling - a pairing of delicacy and grace. Popular in classic and vintage designs.
Important: pearls are organic, soft (2.5-4.5 on Mohs), requiring gentle care.
Cubic zirconia (Synthetic diamonds):
Cubic zirconia is an inexpensive but striking imitation of diamonds. In sterling, they look expensive, creating a "diamond" look for a fraction of the price.
Stones That Look Better in Gold
Some stones traditionally look better in gold:
Ruby, sapphire (except pale blue): The warm glow of gold enhances the depth of colour.
Emerald: Though emerald in sterling is beautiful, the classic pairing is with gold.
Amber: Warm, sunny amber requires a warm metal - gold.
However: These are traditions, not rigid rules. In contemporary design, any pairing is acceptable if it works visually.
How to Test Silver at Home: Simple Methods
Doubtful about a piece's authenticity? There are a few simple home tests. For a deeper walkthrough with photos and edge cases, see the full guide to telling real silver from fake.
Important: No home test offers a 100% guarantee. For absolute certainty - consult a jeweller or assay office.
Method 1: Magnet
Principle: The metal is diamagnetic. It is NOT attracted to a magnet.
Process:
- Take a strong magnet (neodymium; sold in hardware shops)
- Bring the magnet close to the jewellery
- If it is attracted - it is NOT genuine (most likely steel or a nickel alloy with plating)
- If NOT attracted - a good sign, but not a guarantee (many metals are non-magnetic)
Important: This test rules out crude fakes (steel under plating) but does not confirm the metal at 100% (aluminium, copper, gold are also non-magnetic).
Method 2: Thermal Conductivity
Principle: The metal has the highest thermal conductivity of all metals. It instantly takes on the ambient temperature.
Process:
- Place the piece on a table, wait a minute (let it reach room temperature)
- Pick it up, squeeze in your fist
- Sterling will warm to hand temperature in a few seconds
- Fakes (especially plastic with plating) warm more slowly
Or:
- Place an ice cube on the jewellery
- The piece will cool instantly, the ice will begin melting quickly (heat transfer)
However: Copper also has high thermal conductivity (though lower). This test is not 100% reliable.
Method 3: Sound
Principle: The metal, when dropped, produces a pure, long, ringing sound.
Process:
- Drop a ring or coin onto a hard surface (table, tile)
- Listen to the sound
- Genuine piece - a pure, high-pitched, long-lasting ring
- Fakes - a dull, short sound
However: This method is subjective, requires experience. Moreover, it is not applicable to chains and earrings.
Method 4: Acid Test (Reagent)
Principle: A special acid reacts with the metal; the colour of the reaction indicates the fineness.
Where to buy: Jewellery shops, online.
Process:
- Find an inconspicuous spot on the piece (inner surface, reverse)
- Apply a drop of acid
- Observe the colour:
- Bright red → 925 fineness silver
- Dark red → lower fineness silver
- Blue/green → copper, brass
- No reaction → gold or another noble metal
Important:
- The acid etches the surface (leaves a mark). Use only on inconspicuous spots.
- Dangerous (acid!). Use gloves, ensure ventilation.
- Not 100% precise, but fairly reliable.
Method 5: Visual Inspection
Examine the hallmark:
Under a loupe. A quality hallmark is sharp, deep, even. A fake is blurred, crooked, unclear.
Examine the quality:
Genuine 925 from a serious manufacturer means quality workmanship. Flawless polishing, even lines, symmetry.
Crude fakes are usually low quality - uneven soldering, poor polishing, asymmetry.
Tarnishing:
If a piece is presented as genuine, is not rhodium-plated, but does not tarnish for years - it is very likely not authentic.
When to Consult a Professional
If you have:
- Spent a significant sum on the piece
- Doubts about its authenticity
- Purchased an antique or second-hand piece
It is better to pay for a professional assay than to take the risk.
A jeweller will determine the fineness within minutes using:
- Precise acid tests
- Electronic testers (measuring density, conductivity)
- X-ray fluorescence analysis (99%+ accuracy)
925 Silver vs Other Finenesses: Comparative Table
| Characteristic | 925 Fineness | 875 Fineness | 800 Fineness | 999 Fineness |
|---|---|---|---|---|
| Silver content | 92.5% | 87.5% | 80% | 99.9% |
| Hardness | Medium | Above average | High | Low |
| Wear resistance | Good | Very good | Excellent | Poor |
| Tarnishing tendency | Medium | Higher | High | Low |
| Lustre | Bright | Less bright | Duller | Brightest |
| Use | Jewellery | Tableware | Industry | Investment, industry |
| Price | Medium | Lower | Lower | Higher (for purity) |
| For daily wear | Ideal | Good | Acceptable | Unsuitable |
Conclusions:
- 925 - the optimal balance for jewellery
- 875 and 800 - more copper, more durable, but darker and less noble in colour
- 999 - too soft for jewellery, only for investment and industry
FAQ
What is the difference between sterling silver and pure silver? Sterling silver (925) contains 92.5% pure silver mixed with 7.5% copper or other metals. Pure silver (999) is 99.9% silver, which sounds better on paper but is actually too soft for jewellery. It bends, scratches, and loses shape quickly. Sterling silver gives you that beautiful silver look with enough strength to wear every day.
How do I stop my silver jewellery from tarnishing? Tarnishing happens when silver reacts with sulphur in the air, not because something is wrong with your piece. Store your jewellery in airtight bags or lined boxes when you are not wearing it. Keep it away from perfume, hairspray, and cleaning products. Wearing your silver regularly actually helps too, since the natural oils on your skin slow down the tarnishing process.
Can silver jewellery cause allergic reactions? Sterling silver is generally very well tolerated, even by people with sensitive skin. Most silver allergies are actually reactions to nickel, which some lower-quality alloys contain. Look for nickel-free 925 silver to be safe. If you have known metal sensitivities, rhodium-plated silver is an excellent option since it creates a protective barrier between the metal and your skin.
What is the best way to clean silver jewellery at home? Warm water with a drop of mild dish soap and a soft cloth works perfectly for regular cleaning. For tarnished pieces, a paste of baking soda and water applied gently with a soft toothbrush does wonders. Rinse thoroughly and dry completely before storing. Avoid harsh chemicals and abrasive materials, they can scratch the surface and damage any gemstone settings.
Is silver better than white gold for everyday jewellery? It depends on your priorities. Silver is significantly more affordable and has a bright, cool tone that many people prefer. White gold is harder and more scratch-resistant, but it requires rhodium replating every few years to maintain its colour. Silver does tarnish, but cleaning it is simple and inexpensive. For trendy pieces you might rotate often, silver makes more sense financially.
How long does silver plating last? Silver plating on base metals typically lasts anywhere from six months to two years, depending on how often you wear the piece and how you care for it. High-friction areas like ring bands wear through faster. Once the plating wears off, the base metal underneath may cause skin reactions or discolouration. Solid sterling silver avoids this problem entirely, which is why it is a better long-term investment.
Can I wear my silver jewellery every day? Absolutely. Sterling silver is designed to be worn, not locked away. Remove it before swimming in chlorinated pools, using harsh cleaning agents, or exercising heavily. Shower water alone will not hurt it, though soap buildup over time can dull the shine. A quick wipe with a soft cloth after wearing keeps your pieces looking fresh for years.
Does silver work well with gemstones? Silver and gemstones are a natural match. The cool tone of silver beautifully complements blue and purple stones like amethyst, topaz, and sapphire. It also pairs wonderfully with turquoise, moonstone, and pearls. The lower cost of silver compared to gold means you can choose larger or higher-quality stones for the same budget, which often makes the whole piece more striking.
Conclusion: 925 Silver - The Connoisseur's Choice
925 silver is not a compromise, not a "budget alternative to gold." It is a self-sufficient, complete noble material with an extraordinarily rich history and unique properties.
Why 925 Silver Is the Choice of People with Taste
Aesthetics:
Silver's cool, pure lustre represents modernity, freshness, relevance. In the age of minimalism and functional design, silver proves a more "fitting" choice than ostentatious gold.
Versatility:
Silver suits everyone - regardless of age, gender, style, occasion. It is a universal language of elegance.
Accessibility:
Silver democratises luxury. Quality jewellery in a noble metal is accessible to a wide public.
Scope for experimentation:
Silver's accessible price allows one to try different styles, change looks, build extensive collections. It is freedom of expression.
Historicity:
Silver has been valued by humanity for millennia. It features in the myths, religions and cultures of every civilisation. To wear silver is to be part of that history.
Care - Part of Ownership
Yes, silver requires care. It tarnishes; it needs cleaning. But this is not a flaw - it is a characteristic that makes silver "alive."
The ritual of cleaning silver is a moment of attention to one's possessions, a moment of care. In an age of disposability and fast consumption, this has value.
Invest in Quality
Not all 925 silver is equal. Cheap mass production may have the correct fineness but appalling quality of execution.
Choose:
- Trusted brands
- Jewellers with a portfolio and reputation
- Pieces with clear hallmarks
- Quality polishing and detail
Better one quality piece than ten cheap ones.
Silver and gold jewellery, wedding bands, symbolic pendants, paired sets.
Zevira: Silver with Character
Zevira offers 925 silver jewellery created for those who value individuality.
We do not make faceless mass-market goods. Each of our collections is a story, a character, a statement.
What makes Zevira special:
- Only certified 925 silver with the correct composition (nickel-free)
- Design inspired by a passion for craftsmanship - bold, vibrant, yet elegant
- Attention to detail - flawless polishing, considered ergonomics
- Durability - we design jewellery to be worn, not to sit in a display case
Our pieces are not about "cheap and plenty." They are about "quality and character."
If you are looking for silver jewellery that reflects your personality, that withstands years of wear and never goes out of style - welcome to the world of Zevira.
Explore the Zevira collection →
Care for your silver with love. And it will accompany you for decades, reflecting your style and character.
#SterlingSilver925 #SterlingSilver #Zevira #SilverJewellery #SilverCare #925Silver #QualityJewellery #ModernJewellery #RhodiumPlatedSilver #JewelleryGuide